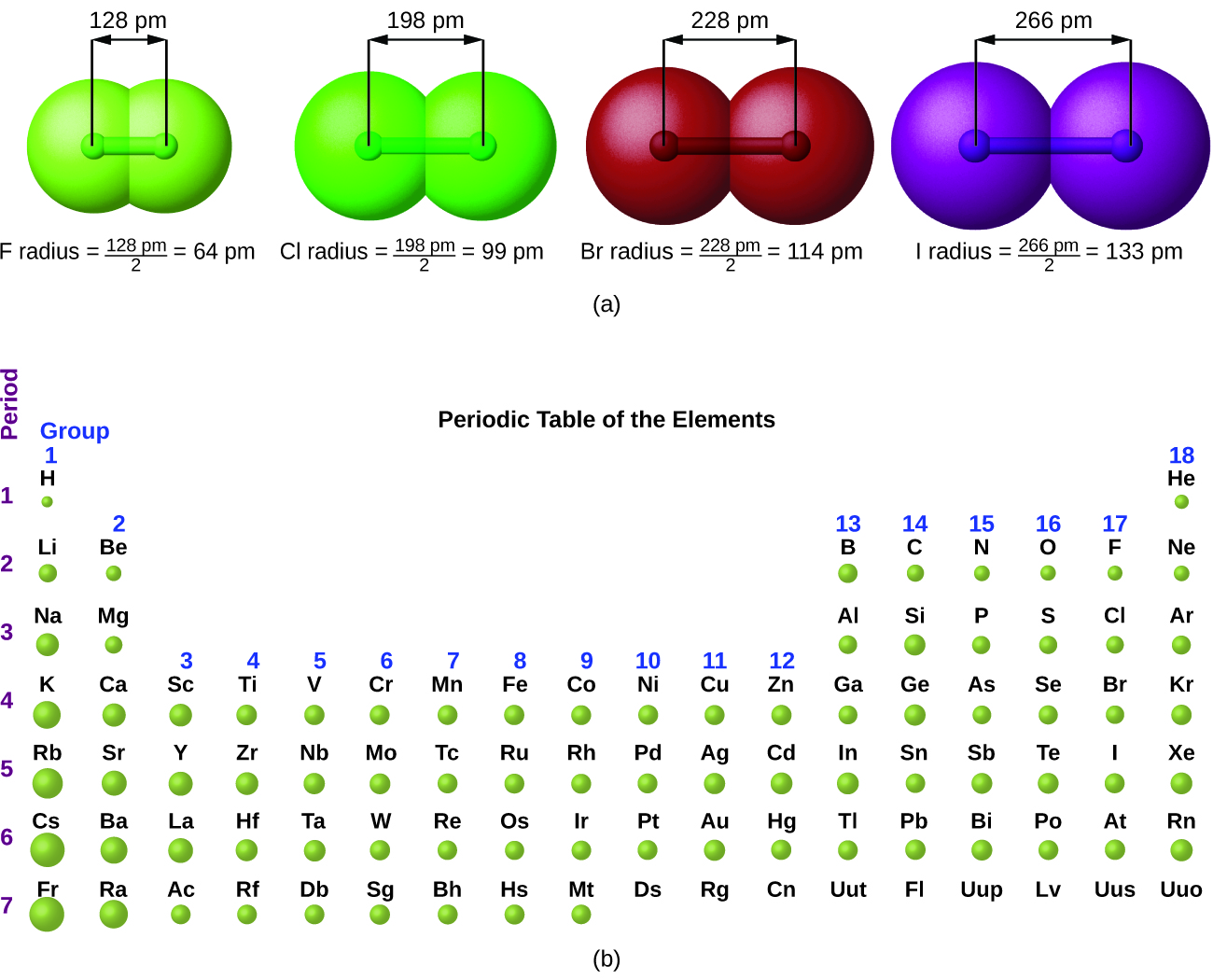
The barium cation is written Ba 2+, not Ba +2. Note the convention of first writing the number and then the sign on a multiply charged ion. Figure 3.3 “Predicting Ionic Charges” shows how the charge on many ions can be predicted by the location of an element on the periodic table. On the other side of the periodic table, the next-to-last column, the halogens, form ions having a 1− charge. Ions made from alkaline earth metals, the second group on the periodic table, have a 2+ charge. For example, all ions made from alkali metals, the first column on the periodic table, have a 1+ charge. Thus, the periodic table becomes a tool for remembering the charges on many ions. In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge because they have the same number of valence electrons. In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. Notice that there are no leftover electrons. The resulting combination is the compound sodium chloride. 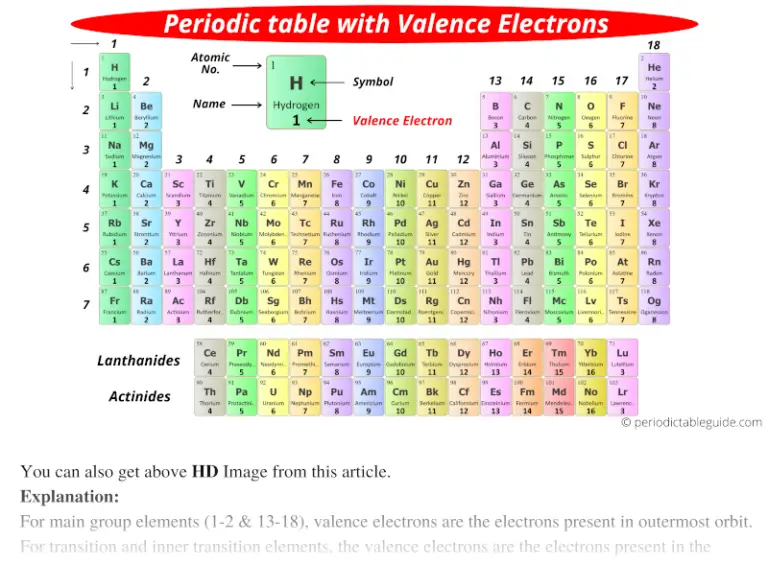
With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. On the right, the chloride ion has 18 electrons and has a 1− charge. Most metals on the periodic table (located on the left of the periodic table) are atoms that will give up their valence electrons from their outermost energy. 
Most nonmetals become anions when they make ionic compounds.įigure 3.2 The Formation of a Chlorine Ion. On the left, the chlorine atom has 17 electrons. Negatively charged ions are called anions. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. Most metals become cations when they make ionic compounds. Positively charged ions are called cations. Atoms that lose electrons acquire a positive charge as a result because they are left with fewer negatively charged electrons to balance the positive charges of the protons in the nucleus. In cases where an atom has three or fewer valence electrons, the atom may lose those valence electrons quite easily until what remains is a lower shell that contains an octet. The vertical columns (groups) of the periodic table are arranged such that all its elements have the same number of valence electrons. Some atoms have only a few electrons in their outer shell, while some atoms lack only one or two electrons to have an octet. The chemical formula of magnesium chloride is MgCl 2.Most atoms do not have eight electrons in their valence electron shell. Similarly, when magnesium cations, Mg 2 +, combine with chloride anions, Cl −, there must be twice as many chloride anions as magnesium cations in order for the charges to balance each other. Note that charges do not appear in the chemical formulas of compounds: you don’t write Mg 2 +O 2 −). This reveals another corner chemists cut when writing formulas. Thus, when Mg 2 +and O 2 − combine to form the ionic compound magnesium oxide, there have to be equal numbers of magnesium cations and oxide anions. And recall when oppositely charged ions combine to form compounds, there must be the same number of positive and negative charges, so that the compound carries no overall charge. However, you can predict whether an atom will form negative or positive ions from its position in the Periodic Table: elements to the left (metals) will tend to form positive ions those on the right (non-metals) will tend to form negative ions. Figure 7.9 Lewis symbols illustrating the number of. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 7.9 shows the Lewis symbols for the elements of the third period of the periodic table. Note that the valency does not indicate whether an ion will carry a positive or negative charge: oxygen, O, also has a valency of two, and forms negative ions, O 2 −. We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. Take for example, magnesium, Mg, this has a valency of two and tends to form ions carrying two positive charges, Mg 2+. The relationship between valence electrons and charge of an ion is EXTREMELY IMPORTANT. In ionic compounds, valency gives an indication of the charge an ion formed from a particular element will carry. 
3.3 Valency and predicting formulas in ionic compounds
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
